American biotech’s biggest problem is not biology
America, China, and the Race to Pharma Sovereignty
The core problem for biotech and pharma innovation today is not scientific ambition, alone. It is a growing efficiency gap in how drugs are developed.
Last week, Panoptic Bio was mentioned in Forbes, in an article on U.S. and Chinese biotech innovation. The article described how China has quietly become biotech’s “back office” through its growing influence in molecule synthesis, CRO execution, and cross-border licensing.
The West will not “science its way” to biotech leadership unless it rebuilds the drug development engine with the same intensity applied to discovery. China’s rise shows what happens when execution capacity compounds.
The United States still leads in frontier AI and foundational science. But without major gains efficiency, that advantage will not translate into durable health leadership or medical sovereignty.
At the center of the efficiency gap is decision intelligence.
The Problem Is Not Just Biology
The headline numbers are familiar, which is why they are dangerous.
Drug development now averages roughly $2.6 billion per approved drug in capitalized cost, based on Tufts CSDD estimates. Time from first-in-human to approval averages about 10.5 years, according to BIO and Informa.
These figures imply something uncomfortable. Even with perfect biology, the system would remain slow and expensive. Too much failure and capital loss is created by how decisions are made.
The industry has pushed hard on discovery. AI for Science and AI for Bio are now standard. Generative design, target discovery, structure prediction, and self-driving labs have advanced quickly. But better candidates do not guarantee better outcomes.
Discovery gains are routinely lost when diligence is manual, portfolio decisions lag data, trials are slow to design, competitive intelligence is fragmented, endpoints are poorly specified, sites under-enroll, protocol amendments multiply, and regulatory strategy becomes reactive.
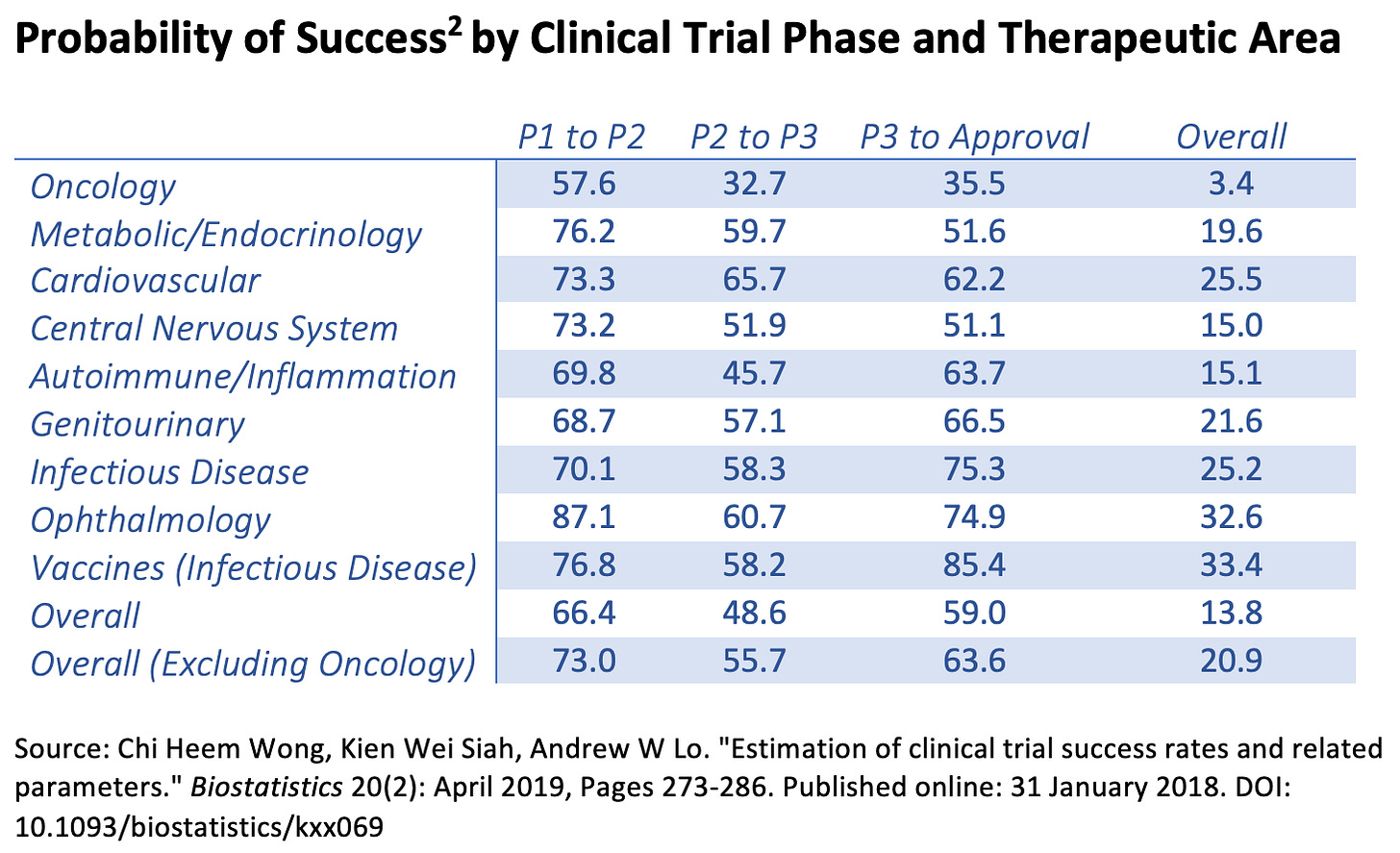
BIO and Informa data show where this breaks down. The probability of a Phase I asset reaching approval is roughly 7.9 percent across 2011 to 2020.
Phase II is the largest bottleneck. Only about 28.9 percent of candidates advance. That is not just biology. It reflects late, noisy learning.
Discovery AI increases the number of bets. It does not ensure better bets. The West still runs programs through slow, fragmented decision systems. The result is not only execution risk. It is low decision quality and the resulting probability of success variability.
China’s Advantage Is Fast Learning
Calling China biotech’s “back office” understates the shift. China did not just build molecules. It built the industrial throughput layer of drug development. Over time, that layer became strategic leverage.
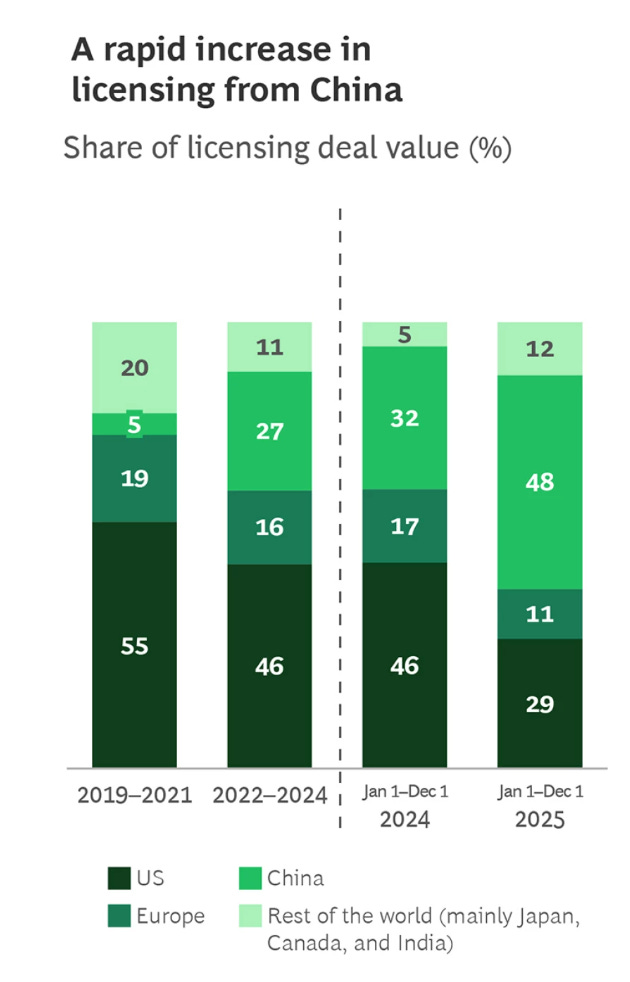
The licensing data is clear. A Boston Consulting Group (BCG) reports that deals involving China drove almost half of global biopharma licensing activity in 2025. Valuations of Chinese assets rose roughly 150 percent year over year, outpacing both US and European peers.
Boston Consulting Group (BCG) analysis on pharma deal value reveals China’s rise
China’s share of global biopharma licensing value grew from about 5 percent in 2021 to roughly 40 to 50 percent by 2025.
In January 2026, AstraZeneca signed a multi-program obesity deal with CSPC valued at up to $18.5 billion, including a $1.2 billion upfront payment. It was one of the largest licensing deals of the 2025 to 2026 period.
China is no longer just lower-cost execution. It is a source of assets and a high-throughput development environment that global pharma depends on.
This advantage came from sustained investment in the execution stack. Large-scale CRO and CDMO capacity turned scientific intent into executable trials. Firms adapted under geopolitical pressure by restructuring and focusing on core platforms. That is industrial maturity.
The real compounding advantage is cycle time. Cycle time determines when decisions are made.
Faster iteration means earlier learning, faster termination of weak programs, tighter capital concentration, and stronger partner pull. This is how a back office becomes a front office.
At its core, China built a fast learning system.
Sovereignty Depends on Decision Loops
The West has already seen efficiency collide with sovereignty in consumer technology. TikTok triggered years of political conflict in the US. China blocked US social platforms much earlier.
Pharma has had its own preview. COVID made medical sovereignty tangible. Countries with domestic development and manufacturing capacity moved first. Others waited.
If algorithms and chips are treated as sovereignty concerns, medicines will be too. Possibly more so.
Medical sovereignty is not only about factories. It is about decision autonomy. The ability to design trials, interpret data, adjust strategy, and advance or kill programs without dependence.
Decision loops are sovereignty.
Collaboration Will Persist
Decoupling is not clean. US-China biopharma collaboration remains deep and, in some areas, growing.
Pharma needs assets. Biotech needs capital and commercialization. Patients need therapies. These pressures are structural.
Pharma faces a patent cliff. Populations are aging. Healthcare demand is rising. Biotech has faced a funding winter since 2021.
Deal data reflects this reality. China’s rising licensing share is proof.
Two things are true. Innovation is global, and collaboration will continue. At the same time, the US and Europe want operational self-sufficiency in critical medicines.
This tension defines the decade. And it leads back to efficiency.
Efficiency Starts with Decision Intelligence
Start with the baseline. $2.6 billion. 10.5 years. 7.9 percent approval probability.
Under these constraints, advantage will not come from discovery alone. It will come from better decision intelligence paired with strong execution.
Discovery creates options. Decision intelligence determines which options deserve capital, time, and operational focus.
The real levers now are earlier selection of the winning 8 percent, real-time integration of biological, clinical, regulatory, and commercial signals, shorter decision cycles, faster trial design, higher signal-to-noise in endpoints, and tighter control of sites and amendments.
AI must extend beyond discovery. Models can propose molecules. The larger prize is compressing and de-risking the clinical and regulatory arc, where most capital is lost.
Most capital is not lost because molecules fail. It is lost because failure is discovered too late.
Turning the US AI Edge Into Pharma Outcomes
The United States retains major advantages. Frontier AI research. Large-scale compute. Deep capital markets. Leading academic medical centers. Influential regulators.
But advantages only matter if they are operationalized. They transfer through tools and workflows that improve everyday decisions.
AlphaFold mattered because it collapsed iteration time and reshaped workflows. The same must happen in clinical development.
AI must become infrastructure. Systems that continuously synthesize evidence and guide decisions in real time.
Given China’s efficiency gains, the US does not have time to wait.
Why Panoptic Bio Exists
This is the gap Panoptic Bio addresses.
If efficiency is the constraint, and Phase I to approval remains a roughly 7.9 percent outcome over more than a decade, the biggest leverage is not marginally better discovery. It is a better clinical-stage decision system.
Faster decision cycles. Higher-quality evidence synthesis. Greater consistency. Scalable learning across programs.
Clinical trials should not be driven by fragmented, manual intelligence. They should be guided by systems that learn continuously from trial reality and translate that learning into better decisions and better execution.
This is where failure is most expensive. It is also where decision intelligence matters most.
If China’s advantage is execution throughput, the Western response must be decision superiority paired with execution strength. That means building a modern pharma operating system that converts AI leadership into sustained biotech leadership.
Panoptic Bio’s Trial Terminal is a decision intelligence platform to safely run AI models and agents on ongoing clinical trials
Retrieve any registered clinical trial from the U.S., Europe, China, or globally
Run Probability of Success analysis on ongoing clinical trials
The Leadership Question
Biotech has long told itself that biology is the bottleneck.
Biology is hard. But the numbers show another constraint. How decisions are made.
China’s rise from back office to strategic center is both warning and lesson. Execution compounds. But execution follows decisions.
If the West wants to lead scientifically, commercially, and geopolitically, it must compete where outcomes are determined.
Decision intelligence is now the frontier.
Sources
Panoptic Bio: https://www.panoptic.bio
Forbes: https://www.forbes.com/sites/shimiteobialo/2026/01/21/how-china-became-the-biotech-industrys-back-office/
BCG: https://www.bcg.com/publications/2026/reimagining-business-models-biopharma-trends
GlobalData: https://www.globaldata.com/media/business-fundamentals/large-pharma-drug-licensing-from-china-reaches-record-high-at-28-in-2024-reveals-globaldata/