The Delivery Gap in Biotechnology
Introducing Our Trial Pipeline Intelligence Series
Biology is moving faster than ever. We can edit genes, engineer cells, and reprogram the immune system with a level of precision that would have sounded like science fiction twenty years ago. The technical barriers that once defined biotechnology are falling one by one.
And yet, therapies stall, and will continue to stall, in clinical trials.
Not because the science fails, but because delivery to patients fails.
Today, we are launching a new series analyzing clinical trial pipelines in the disease areas we believe matter most for the future of medicine and the world. We will periodically publish deep, data-driven assessments.
On www.panoptic.bio we will publish analysis we believe is critical for the public domain.
The first 5 public analyses will be: CNS and neurodegeneration, psychiatry, oncology, and infectious disease preparedness.
These are not random categories. They are the frontiers where human suffering is enormous, scientific progress is accelerating, clinical risk remains poorly quantified, and capital allocation ultimately determines outcomes.
We believe the central bottleneck in modern biotechnology is no longer target discovery alone. It is clinical translation.
Our public assessment of Cell and Gene Therapy (CGT)
We are publishing today an analysis of the global CGT pipeline using Panoptic Bio’s clinical trial intelligence.
We begin this series with Cell and Gene Therapy because it is the clearest example of the delivery gap in modern biotechnology. Scientific feasibility has been crossed. Clinical friction remains severe. The economic upside is enormous. Infrastructure maturity is uneven.
In this inaugural public report, we analyze active private-sector CGT trials across five disease domains, benchmark predicted phase transition probabilities against historical baselines, quantify domain-level pipeline robustness using our alpha score, and identify where clinical friction suppresses value creation. Some domains exceed historical expectations. Others fall materially below.
The story is not uniformly optimistic, and that is precisely why it matters.
This series is our attempt to make clinical translation measurable, comparable, and transparent. Our intelligence is generalizable across disease areas and drug modalities. The same framework applies to small molecules, biologics, cell therapies, gene therapies, neuropsychiatric programs, oncology platforms, and vaccine pipelines.
If biotechnology is to deliver on its promise, we must measure what matters. Clinical translation is measurable. Risk is quantifiable. Pipeline strength is comparable.
The first installment, Cell and Gene Therapy, is live.
You can find it here: www.panoptic.bio/intelligence/cgt
Why We Care About These Frontiers
These disease areas share three defining characteristics.
First, they represent massive human burden. Chronic disease costs the United States more than $1.1 trillion annually in direct care alone. Globally, hundreds of millions of people live with genetic disorders that are, in principle, now biologically addressable. Neurodegeneration erodes identity. Psychiatric illness reshapes lives silently and pervasively. Oncology still claims millions of lives each year. Pandemics can destabilize civilization in months. These are not niche markets. They are civilizational challenges.
Second, they are technologically inflecting. In cell and gene therapy, the science is no longer hypothetical. Dozens of CGTs have already received FDA approval, and more than 3,000 CGT trials are currently underway globally. CAR-T therapies have achieved remission rates approaching 90% in certain leukemias, and CRISPR-based editing has reduced sickle cell pain crises by 97%, offering what is effectively a functional cure for many patients. The breakthroughs are real. The capabilities are tangible. But the pipeline remains fragile.
Third, clinical trials are the bottleneck. Despite representing a small fraction of total trials, CGT programs account for a disproportionate share of FDA clinical holds. This is the delivery gap. Biology is accelerating; industrialization is not. Across modalities including CGT, CNS therapeutics, oncology, and psychiatry, the fundamental constraint is the same: we do not quantify trial-level probability of success rigorously enough. Capital gets allocated based on narrative. Risk is underpriced. Failure compounds. This is not an unsolvable problem. It is a measurement problem.
How We Do Our Analysis
At Panoptic Bio, we focus on clinical trial intelligence. Our models do not simply rank drugs; they estimate calibrated probabilities of phase transition success from Phase I to II, II to III, and III to approval. We analyze active private-sector trials, match each to disease- and phase-specific historical benchmarks, and estimate Probability of Success in a way that is directly comparable to precedent. We quantify uncertainty using 95% confidence intervals and aggregate trial-level predictions into domain-level pipeline robustness scores.
The underlying model is trained on 11,600 trials and more than 560,000 documents. It achieves strong benchmark performance and produces calibrated probabilities with explicit uncertainty estimates. This allows us to move from vague impressions such as “this looks promising” to quantitative statements such as “this program is 20% above historical disease- and phase-matched benchmarks,” or conversely, “this pipeline is structurally below precedent.” That distinction matters for founders making go or no-go decisions, for investors constructing portfolios, for pharmaceutical companies allocating capital, and for policymakers shaping regulatory frameworks.
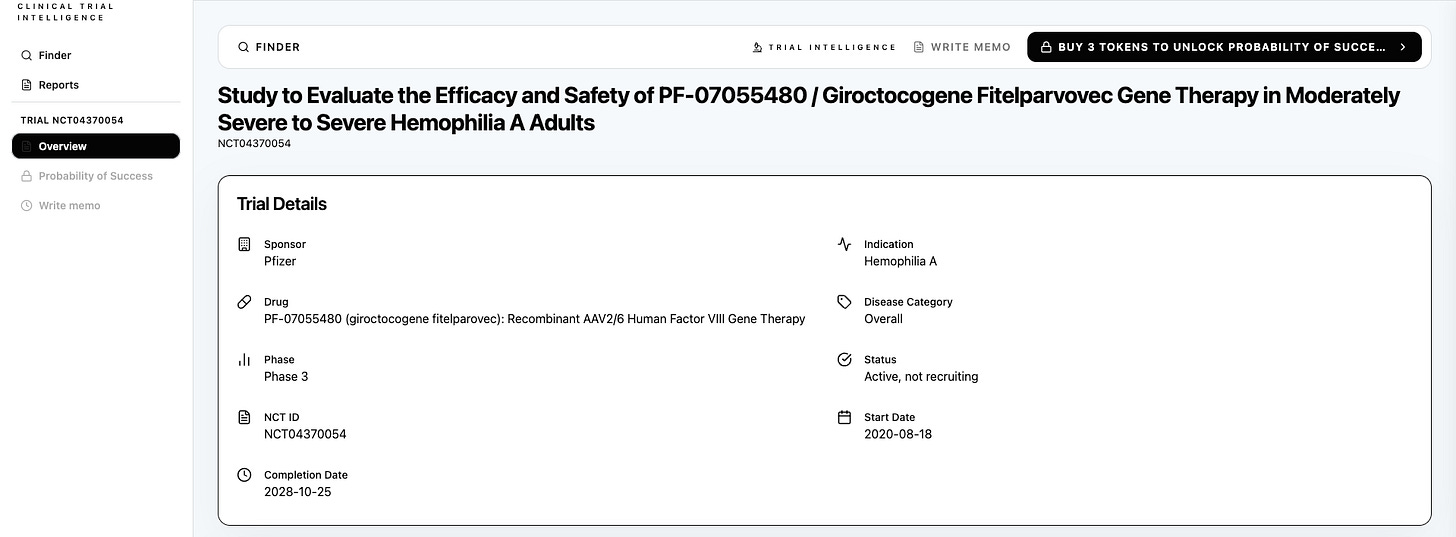
Retrieve any registered clinical trial that is ongoing for probability of success analysis with Panoptic Bio. Shown here: Gene Therapy for Hemophilia A, a clinical trial by Pfizer. Panoptic Bio’s clinical trial intelligence shows an above-benchmark Probability of Success from Phase 3 to market for Pfizer’s gene therapy, with the potential to serve more than 300,000 patients globally living with Hemophilia A.
Why This Matters
Clinical trial efficiency is not merely a scientific, operational, and financial question. It is a moral one.
For example, cell and gene therapy expands into large-population diseases, it represents a serviceable addressable market of roughly $180 billion. But more importantly, it represents a structural shift from managing chronic disease to resolving it. Every failed trial burns capital, delays cures, distorts signal, and increases skepticism across the ecosystem. Poorly calibrated risk compounds systemic inefficiency.
Better clinical intelligence can improve portfolio construction, identify structurally strong domains, flag fragile pipelines early, inform regulatory reform, and accelerate the deployment of curative therapies. The science already exists. The infrastructure for translating it must catch up.
Panoptic Bio is a pharma intelligence company based in Mountain View, CA. Shown here the www.panoptic.bio website with several pharma agents for hire, the Trial Terminal, and public intelligence reporting available.